 The number in a mole is called Avogadro’s number : 6.022142 x 10 23, after the 19th-century Italian scientist who first proposed how to measure the number of molecules in a gas. Just as 1 mol of atoms contains 6.022 × 10 23 atoms, 1 mol of eggs contains 6.022 × 10 23 eggs. According to the most recent experimental measurements, this mass of carbon-12 contains 6.022142 × 10 23 atoms, but for most purposes 6.022 × 10 23 provides an adequate number of significant figures. The mole is used for this purpose.Ī mole is defined as the amount of a substance that contains the number of carbon atoms in exactly 12 g of isotopically pure carbon-12. Any readily measurable mass of an element or compound contains an extraordinarily large number of atoms, molecules, or ions, so an extraordinarily large numerical unit is needed to count them. Atoms are so small, however, that even 500 atoms are too small to see or measure by most common techniques. Sheets of printer paper are packaged in reams of 500, a seemingly large number. 
For example, cans of soda come in a six-pack, eggs are sold by the dozen (12), and pencils often come in a gross (12 dozen, or 144). Many familiar items are sold in numerical quantities that have unusual names. The quantity of a substance that contains the same number of units (e.g., atoms or molecules) as the number of carbon atoms in exactly 12 g of isotopically pure carbon-12., from the Latin moles, meaning “pile” or “heap” ( not from the small subterranean animal!). The unit that provides this link is the mole (mol). To analyze the transformations that occur between individual atoms or molecules in a chemical reaction it is therefore absolutely essential for chemists to know how many atoms or molecules are contained in a measurable quantity in the laboratory-a given mass of sample. In the laboratory, for example, the masses of compounds and elements used by chemists typically range from milligrams to grams, while in industry, chemicals are bought and sold in kilograms and tons. Because the masses of individual atoms are so minuscule (on the order of 10 −23 g/atom), chemists do not measure the mass of individual atoms or molecules. The problem for Dalton and other early chemists was to discover the quantitative relationship between the number of atoms in a chemical substance and its mass. 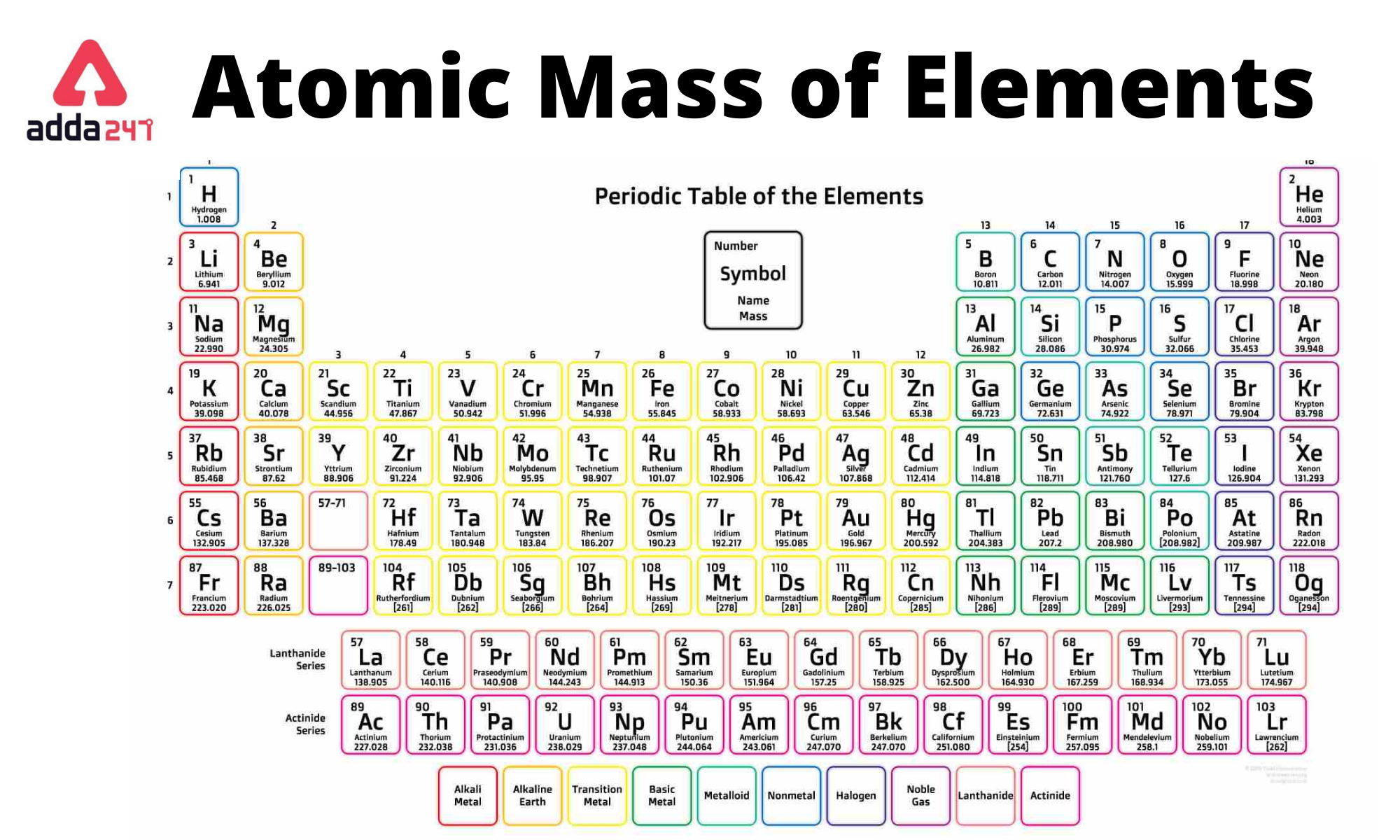
We also described the law of multiple proportions, which states that the ratios of the masses of elements that form a series of compounds are small whole numbers. In Dalton’s theory each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. The same calculation can also be done in a tabular format, which is especially helpful for more complex molecules: To use this calculator Click Here.\right ) \right ] \) To use this resource Click Here.Ī useful resource, written by Jef Rozenski, calculates all possible combinations of H, C, N & O that give a specific nominal mass. Of course, compounds of chlorine and bromine have very large isotope abundances.Ī more elegant and complete isotope pattern calculator has been created by Junhau Yan. The numbers displayed in the M+1 and M+2 boxes are relative to M being set at 100%. Simply enter an appropriate subscript number to the right of each symbol, leaving those elements not present blank, and press the " Calculate" button. The calculator on the left may be used to calculate the isotope contributions to ion abundances 1 and 2 amu greater than the molecular ion (M). For compounds of chlorine and bromine, increments of 1.997 and 1.998 respectively must be added for each halogen to arrive at the higher mass isotope values. Only the mass of the most abundant isotope, relative to C (12.0000), is used for these calculations. The mass calculator on the right may be used to calculate the exact mass of a molecule based on its elemental composition. 
This table is adapted from Introduction to Mass Spectrometry, by J.T. The factor is multiplied by the number of atoms (n) of the designated element to calculate the intensity contribution from higher mass isotopes. * X represents the relative intensity of the lowest mass ionin an isotopic ion cluster. Masses Exact Masses & Isotope Abundance Ratios Element 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
